|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
Short Communication |
||||
|
- |
||||
|
Volume 24 |
April - June 2008 (Part-II) |
Number 3 |
||
|
|
||||
|
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
Short Communication |
||||
|
- |
||||
|
Volume 24 |
April - June 2008 (Part-II) |
Number 3 |
||
|
|
||||
|
|
||||
Study of Alkaline phosphatase from
human hydatidiform mole
Mohammad Aberomand1, Faker Rahim2, Seyed Ahmad Hosseini3
ABSTRACT
Objective: Study was performed on purification of alkaline phosphatase from Hydatiform mole.
Methodology: The sample of mole Hydatidiform subjects from Taleghani Hospital, Tehran, Iran was studied. An Alkaline phosphatase from Human Hydatidiform mole was purified by a protocol involving solubilization using precipitation by butanol, acetone, Ammonium sulphate, Sephadex G200, Ion exchange chromatography and preparative electrophoresis .
Results: The enzyme was purified 800-fold to apparent homogeneity. This enzyme has 5.2% carbohyrate content. The optimum Temperature and pH were 40 centigrade degree and 10.4, respectively.
Conclusion: Human Hydatidiform mole alkaline phosphatase is a novel alkaline phosphatase.
KEY WORDS: Alkaline Phosphatase, Hydatidiform mole, Ion exchange.
Pak J Med Sci April - June 2008 (Part-II) Vol. 24 No. 3 471-474
How to cite this article:
Aberomand M, Rahim F, Hosseini SA. Study of Alkaline phosphatase from human hydatidiform mole. Pak J Med Sci 2008;24(3):471-4.
1. Mohammad Aberomand, Ph.D
Dept. of Biochemistry, School of Medicine,
2. Faker Rahim, M.Sc Bioinformatics
Research Center of Physiology,
3. Seyed Ahmad Hosseini, M.Sc
Department of Nutrition,
1-3: Ahwaz Jondishapur University of Medical Sciences,
Ahwaz – Iran.
Correspondence
Mohammad Aberomand,
Department of Biochemistry,
School of Medicine,
Ahwaz Jondishapour University of Medical Sciences,
Ahwaz – Iran.
E-mail: aberumand@yahoo.com
* Received for Publication: November 14, 2007
* Accepted: April 1, 2008
INTRODUCTION
Alkaline Phosphatase is synthesized by Kidney, liver, bone, Intestine and Placenta.
1,2 Alkaline Phosphatase, at high levels of activity, has long been associated with calcifying tissues; however, its role in the calcification process remains unclear.3 Liver, bone and Intestine Alkaline Phosphatase are the isoenzymes of Ap most commonly found in serum. Human Alkaline Phosphatase (hpALP EC: 3.1.3.1), an isoenzyme of the Alkaline Phosphatase (AP) group of enzymes, ordinary is synthesized in the placenta.4 For alkaline phosphatase synthetic substrate p-nitrophenyl phosphate is used as substrate.1METHODOLOGY
Acrylamide, glycine (Hayashi Pure Chemical Ind. Ltd., Japan); N, N
i-Methylene bisacrylamide and N, N, Ni, Ni-tetramethyl-ethylene diamine (Eastman Organic Chemicals, New York); Sodium dodecylsulphate, ß-mercaptoethanol, ß-Alanine, Polyethylene glycol 6000 (Sisco); CEoomassie Brilliant blue R-250, Sephadex G200, Bio-gel P-100 and Blue dextran 2000, CM-cellulose, (Pharmacia Fine Chemicals, Sweden); Sodium azide (Reidel, Germany); Ammonium per-sulphate (May and Baker Ltd., England); Ethylenediamine tetra acetic acid (E. Merck, Germany); Bromophenol blue.Purification procedures in all operations were carried out at 4°C. The buffer used in the purification procedures was tris buffer (pH 10.5, 0.025 M).
Step I: 100gr of Hydatidiform mole washed and was homogenized in a mixer with saline (0.85% NACl), the homogenate was then mixed with 1-butanol (10ml/50ml of homogenate); the mixture was stirred for one hour at 4
oC and the butanol layer was removed by centrifugation at 12.080g. The treatment was repeated three / four times to ensure complete removal of lipids and pigments.6Step II: The homogenate was mixed with equal amount of cold acetone 0
oC under stirring. The resulting precipitate was collected by centrifugation at 12.080g. The precipitate was extracted with same saline (100ml), with stirring for 5-6 hours at 4oC. The suspension was centrifuged at (12,080g) at 4oC, the supernatant was collected and the residue was re-extracted with saline as before.7Step III: The combined supernatants were pooled and subjected to fractional precipitation with ammonium sulfate, essentially according to the procedure of Agrawal and Goldstein.
8 The protein fraction precipitated between 30%-80% ammonium sulfate saturation was collected by centrifugation at 4oC, dissolved in minimum amount of distilled water, extensively dialyzed against distilled water and finally against saline I. The dialyzed protein solution was centrifuged 12,080g at 4oC and the clear solution containing glycosidase and phosphatase activities (Fraction A) was preserved at -20oC.Step IV: Gel filtration of Fraction A on Sephadex G
200. Fraction A (»20mg protein) was dissolved in 5ml of tris buffer (pH 10.5, 0.1 M) and loaded on a column of Sephadex G200 (2.5×27cm) equilibrated in the same buffer and fractions of 3ml were collected. The fractions were analyzed for protein as well as for enzyme activities. The fractions rich in enzyme activities were pooled together for next step.9Step-V: Ion exchange chromatography of Gel filtration fraction on DEAE cellulose. Alkaline Phosphatase from wash off fraction was further purified by ion exchange chromatography on DEAE cellulose column. Enzyme was eluted with discontinuous gradient of NaCl (0.01-1 M.).
6Step VI: Preparative Electrophoretic analysis. The enzymatically active pools from before step of purification analysed by the PAGE system of Laemmli.
10 The electrophoresis was performed using two parallel continuous 7.5% gels at 4°C. After an electrophoretic run, one gel was stained for protein with Coomassie Brilliant Blue.5,11Protein Concentration and Carbohydrate Content: Protein was quantified by the method of Lowry et al with bovine serum albumin as standard.
12 Protein was also measured by absorbance at 280nm assuming that A280 = 1.0 corresponds to 1mg/ml.13 Phenol sulphuric acid method was used for determination of carbohydrate using glucose as standard.14Estimation of Alkaline phosphatase activity: 200ml of p-nitrophenyl phosphate disodium salt solution (2mM) was incubated with an enzyme solution in the presence of tris buffer (pH 10.5, 0.1 M) in a total volume of 500ml at 30
oC+/- 0.1. The reaction was arrested after suitable time period by addition of 2.5ml borate buffer (pH 9.5, 0.1 M). The liberated p-nitrophenol was monitored spectrophotometrically at 405nm.6Influence of pH on enzyme activities: 100µl of enzyme ( 10 units) was incubated with 200µl of tris buffer of pH ranging between pH 3.0 to pH 6.0 for 1 hour and 200µl p-nitrophenyl substrates were added. Reaction was arrested after suitable time period by addition of 2.5ml of borate buffer (pH 9.5, 0.1M). The released p-nitrophenol was estimated at 405nm.
6Influence of temperature on enzyme activities: 100ěl of enzyme was incubated with 200µl of tris buffer of optimum pH at different temperatures ranging between 0
oC -100oC for 30 min. 200µl of substrate was added. The reaction was arrested after suitable time period by addition of borate buffer (pH 9.5, 0.1M) and released p-nitrophenol was monitored at 405nm.6RESULTS
The first purification step of the crude preparation by gel permeation chromatography gave three peaks. The enzymatic activity of the three peaks was assayed, as well as that of the fraction-A preparation. The fraction-A has prepared from saline extract when subjected to preparative gel filtration on sephadex G
200. However high fold increase in activity was observed (Table-I).
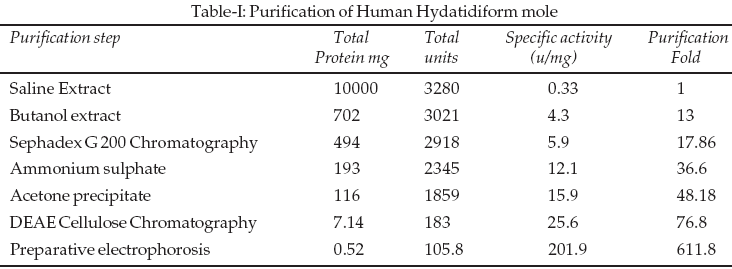
Enzyme activity containing fractions were pooled and used for further purification. Chromatography profile of reach fractions of enzyme of sephadex G200 on DEAE cellulose column, Alkaline phosphatase could be eluted from the column by gradient elution with NaCl concentration gradient between 0.01 and 0.1M was able to separate Alkaline phosphatase. Preparative electrophoresis was chosen as the next step of purification. Alkaline phosphatase and other enzymes could be eluted from the column of gel electrophoresis was able to separate alkaline phosphatase and other enzymes (Figure-1). The purity and homogeneity of the purified enzyme was checked by electrophoresis in native PAGE (pH 8.3). Alkaline phosphatase was further subjected to gel filtration on sephadex G200 when a single peak was obtained for enzyme indicating homogeneity. Enzyme contains 5.2% carbohydrate. With ń-nitrophenyl phosphate in tris buffer as substrate alkaline phosphatase shows maximum activity at pH 10.4 and optimum temperature 40oC.

DISCUSSION
Human Hydatidiform mole alkaline phosphatase has special characterization namely optimum temperature and pH, carbohydrate contain that these characterization are difference with another alkaline phosphatase.
CONCLUSION
Human Hydatidiform mole alkaline phosphatase is a novel alkaline phosphatase.
ACKNOWLEDGMENTS
We would like to express our gratitude to Prof. Bigen Farzami for his guidance, advice, considerate support, encouragement and help throughout the duration of this work. Authors also want to thank Dr. Azar Khosravi for her guidance.
REFERENCES
1. Ohkubo A, Langerman N, Marshal MK. Rat liver alkaline phosphatase. J Biochemi Chemistry 1974;249:1774-80.
2. Terao M, Studer M, Gianni M, Garattini E. Isolation and characterization of the mouse liver/bone/kidney-type alkaline phosphatase gene. Biochem J 1990;268:641-8.
3. Grant WC, Roy EW. Purification and Initial Characterization of Intrinsic Membrane – bound alkaline phosphatase. From Chicken Epiphyseal Cartilage. J Biochem Chemistry 1981;256:7262-8.
4. De GG, De PV, De VA, De Broe MFW. Use of monoclonal antibodies to detected human placental alkaline phosphatase, Clinical Chemistry 1983;29:115- 9.
5. Williams DE, Reisfeld RA. Disc electrophoresis in polyacryamide gel electrophoresis gels: Extension to new conditions of pH and buffer: Annal. N Y Acad Sci 1964;121:373-81.
6. Aberomand M, Farzami B. Purification and Characterization of Alkaline phosphatase from Placental, J Sabzevar School of Med Sci Health Serv 2000;4:32-9.
7. Aberomand M, Soltanzadeh M. Purification and Characterization of Psedocholinesterase from sheep Liver. Biochemical and Cellular Archive 2006;6:253-6.
8. Aberomand M, Bhide SV. Purification and Characterization of novel Mannosidase from Human serum, J Sabzevar School Medical Sciences and Health Services 2007;40:1-10.[article in Persian]
9. Aberomand M, Bhide SV. Purification and Characterization of alpha mannosidase from Chickpea seeds, Biochemical and Cellular Archive 2007;7:242-7.
10. Laemmli UK. Cleavage of Structural proteins during the assembly of the head of bacteriophage T4. Nature 1970;227:680-5.
11. Unakami S, Hirata M, Ichinohe K, Tanimoto Y, Iizuka H. Separation and quantification of serum alkaline phosphatase isozymes in the rat by affinity electrophoresis. Jikken Dobutsu 2006;64:1663-9.
12. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Colorimetric method for determination of sugars and related substances. J Biol Chem 1951;193:265-356.
13. Sawhney SY, Bhide SV. Immobilized Mucin: an affinity matrix for the isolation of winged bean acidic and basic lectins. J Chromtogr 1990;503:2272-765.
14. Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugars and related substances. Anal Chem 1956;28:350-6.
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk