|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 25 |
April - June 2009 (Part-II) |
Number 3 |
||
|
|
||||
|
|
||||
|
|
||||
|
Published by : PROFESSIONAL MEDICAL PUBLICATIONS |
||||
|
ISSN 1681-715X |
||||
|
||||
|
- |
||||
|
ORIGINAL ARTICLE |
||||
|
- |
||||
|
Volume 25 |
April - June 2009 (Part-II) |
Number 3 |
||
|
|
||||
|
|
||||
Effect of Zinc Sulphate on Peptic Ulcer Disease
Kambiz Yazdanpanah1, Nasrin Moghimi2,
Vahid Yousefinejad3,
Ebrahim Ghaderi4, Nazila Darvishi5
ABSTRACT
Objective: In-vitro study, the effect of zinc sulfate on healing ulcers is being shown but it is a limited study which has been done to demonstrate the effect of zinc sulfate on peptic ulcer in human. This study was conducted to investigate the effect of zinc sulfate on healing peptic ulcer disease.
Methodology: This was a double blind randomized clinical trial in which patients who were referred to endoscopy clinic in Sanandaj Besat hospital were included. Patients were allocated to intervention group (n=46) and control group (n=58) at random. All patients received standard treatment for ulcer containing Amoxicillin, Metronidazole, Omeprazole and Bismuth. For intervention group one zinc sulfate capsule (220 mg) was added to treatment regimen (one capsule on alternate day). After four weeks, endoscopy was repeated again. The data was analyzed by SPSS software and x˛, Fisher exact and T test were used for analysis.
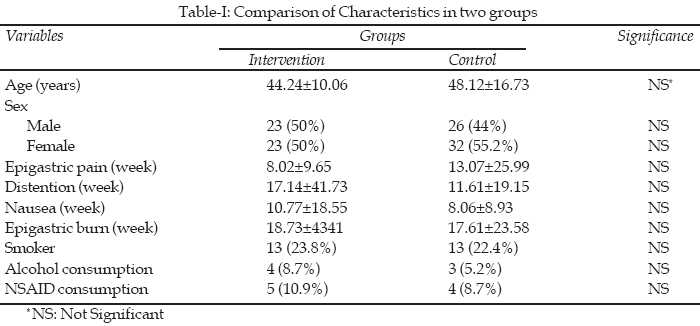
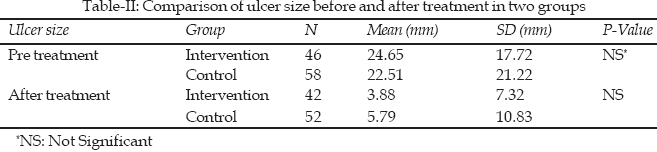
Results: There was no difference between intervention and control group in the same factors like smoking, alcohol, NSAID drugs consumption and the size of ulcer. Mean size of ulcers in intervention and control group before treatment were 24.6 (±17.7)mm and 22.5 (±21.2)mm respectively. The mean percentage size reductions of ulcer in intervention and control group after treatment were 92 (±14) percent and 89.4 (±16.4) percent which did not illustrate significant difference in two groups.
Conclusion: This study confirms that zinc sulfate with the dosage of 220 mg on alternate days doesn’t have significant effect on peptic ulcer healing. More studies with higher doses needs to be undertaken to determine effective dose of zinc.
KEYWORDS: Zinc sulfate, Peptic ulcer, RCT.
Pak J Med Sci April - June 2009 (Part-II) Vol. 25 No. 3 404-407
How to cite this article:
Yazdanpanah K, Moghimi N, Yousefinejad V, Ghaderi E, Darvishi N. Effect of Zinc Sulphate on Peptic Ulcer Disease. Pak J Med Sci 2009;25(3): 404-407.
1. Kambiz Yazdanpanah,
2. Nasrin Moghimi,
1,2: Assistant Professor of Internal Medicine,
Department of Internal Medicine, Besat Hospital,
3. Vahid Yousefinejad,
General Practitioner,
4. Ebrahim Ghaderi,
General Practitioner,
Research Master of Research Deputy,
1-4: Kurdistan University of Medical Sciences,
Sanandaj, Iran.
5. Nazila Darvishi,
General Practitioner, Vahdat Health Care Center,
Sanandaj District Health Center,
Sanandaj, Iran.
Correspondence
Kambiz Yazdanpanah
E-mail: hooman56y@yahoo.com
* Received for Publication: February 9, 2008
* Revision Received: April 27, 2009
* Revision Accepted: May 6, 2009
INTRODUCTION
Peptic ulcer is disruption of duodenum or stomach mucous that leads to local defect and active inflammation. The acid–pepsin disorder is very common in US and involves about 4 million people every year. The prevalence of peptic ulcer is about 12% in men and 10% in women.
1The predisposing factors are H.pylori infection, long term consumption of NSAID and acid over secretion. Other factors like stress, personality type, Alcohol, smoking and diet are also involved. One of the problems in peptic ulcer is healing of the ulcers that could decrease the complications.
2Zinc is important for body metabolism, growth, activation of about 300 enzymes, gastric acid secretion reduction and tissue healing,
3-5 but the effect of zinc on tissue healing isn’t completely known.4 A few clinical and animal studies have showed the effect of zinc components in treatment and prevention of peptic ulcer.6-8 In one study, the zinc serum level of people with peptic ulcer was lower than normal population while zinc amount in gastric mucus was higher probably because of zinc aggregation in injured mucus.9 Zinc deficit in serum and peptic tissue (gastric & duodenum) could show its pathological effect on ulcer, but this is still controversial.10 However studies on animal and human have shown the zinc cytoprotective effect.11-15This study was undertaken to investigate effect of zinc sulfate consumption on healing of peptic ulcer disease.
METHODOLOGY
This study was a double blind randomized clinical trial (RCT). We studied all patients who were referred to endoscopy clinic in Sanandaj Besat Hospital. Out of 164 patients 104 participants fulfilled inclusion criteria and were allocated in intervention (n=46) and control group (n=58) by randomized blocking method.
Inclusion criteria consisted of patient’s satisfaction, peptic ulcer confirmed by endoscopy and no treatment for peptic ulcer during four last weeks. For every person one questionnaire was filled in before endoscopy. Exclusion criteria included evidence of ulcer malignancy in pathology result and patients not willing to go for endoscopy again. An internist did the upper GI endoscopy and treatments were started using four drugs: Amoxicillin 500 mg QID, Metronidazole 500 BID, Omeprazole 20 mg/day and Bismuth 240 mg TDS for all patients. A nurse allocated patients in intervention or placebo group by random blocking method and delivered 220 mg zinc sulfate capsules (every other day) for intervention group.
After four weeks, all patients filled in questionnaire again and endoscopy was repeated. The study was explained to patients and informed consent was taken from participants. The Kurdistan University of medical sciences Ethic Committee approved this study. The data were analyzed by SPSSwin 11.5 and x˛, Fisher, and T test were used for analyzing the data.
RESULTS
Of 104 patients, 46 patients (44.2%) were in intervention group and 58 patients (55.8%) in control group. Four patients in intervention and six in control group did not agree to have follow-up endoscopy. Demographic characteristics and patient’s history have been shown in Table-I. In intervention group, the ulcer size of less than 30 mm was in 32 (69.6%) and more than 30 mm was 14 (30.4%). In the control group 43 ulcer cases(74.1%) were less than 30 mm and 15 cases (25.9%) were more than 30 mm; so there was no significant difference between two groups (p>0.05). The location of ulcers was as follows: 29 (27.9%) ulcers in stomach, 75 (72.1%) in duodenum, four (3.8%) in stomach, 17 (16.3%) in antrum, four (3.8%) in fundus, four (3.8%) in cardia, 70 (67.3%) in first part of duodenum and five (4.8%) in second part of duodenum.
The mean size of ulcers in intervention and control group before treatment were 24.6 (±17.7) mm and 22.5 (±21.2) mm which means there was no significant difference. The mean percentage size reduction of ulcers in intervention and control group after treatment were 92 (±14) and 89.4 (±16.4) percent. There was no significant difference in mean of ulcer size between intervention and control groups, before and after treatment (Table-II).
DISCUSSION
Zinc is an important element for metabolism, growth, and tissue healing
3-5 but, the mechanism of zinc effect on tissue healing isn’t completely clear.4 Some of clinical and laboratory studies have shown the effect of zinc components in treatment and prevention of gastric ulcers.6-8 In one study, the serum level in patients with peptic ulcer was lower than normal population and zinc amount in gastric mucus was higher, that may be caused by zinc accumulation in injured mucus.9 Our results showed that the percentage of reduction of mean ulcer size in intervention group was 92.03 that is higher than control group (89.42%), but there was no significant statistical difference. Perhaps, it may be due to low zinc ulfate dose (220mg/day, every other day).One laboratory study in Hong Kong showed that ulcer healing in rat increased significantly; this process was dose related. They used 88mg/kg zinc which is higher than our dosage. Also another study used 220 mg zinc, tree times a day, and consequently ulcer healing in intervention group was higher three times more than control group.
16 In one animal model study, zinc, with 20 mg/kg dosage, had preventive effect on ulcer.17One study in Spain showed that 600mg/kg dosage had more effect on duodenal ulcer healing comparing with 40mg/kg/day Famotidine.
18 In comparison with other studies, the zinc dosage in this study was lower than the others. This is because of disapproval of ethic committee of Kurdistan University of medical sciences for using higher doses. In our study in intervention group only nausea had increased relatively. Therefore in future studies we could use higher doses of zinc sulfate. On the other hand we didn’t detect the level of serum zinc before the study, so it was a defect in our study. However one study in India showed that serum zinc level in patients with peptic ulcer disease is lower than normal people.9In this study, the ulcer size in both groups had been reduced, and mean ulcer size after treatment in intervention group was less than control group but there was no significant statistical difference between two groups. The mechanism of zinc effect is not known exactly. In one in-vitro study in US the effect of zinc sulfate and acetylcystein on gastric ulcer was confirmed, probably sulfidryl components induced their effects
5 and basal gastric acid secretion decrease was suggesting.19In another study some drugs like zinc sulfate, meciadanol, propranolol, dipiridamol, which in animal studies have cytoprotective effect, have been suggested.
20 One study assessed the effect of zinc sulfate, on ulcers which have been induced by alcohol and indomethacin in rat stomach, and assessed the changes in PGE2 and histamine levels in gastric mucus glands. Alcohol reduces the levels of histamine while indomethacine reduces the PGE2 in gastric mucus glands. Also alcohol increases histamine in gastric secretions. Zinc sulfate reverses the changes of histamine mucosal and secretory levels which are induced by alcohol but has no effect on PGE2 reduction which is induced by indomethacine.7CONCLUSION
This study showed that zinc sulfate in dose of 220 mg/day every other day doesn’t accelerate peptic ulcer healing.
ACKNOWLEDGMENT
We thank Dr. N Moghbel, Mr. S Shahsavari and employers of endoscopy Clinic in Sanandaj Besat hospital for their co-operations.
REFERENCES
1. Del Valle J. Peptic ulcer disease and related disorders. In: Kasper DL, Braunwald E, Fauci AS, Hauser SL, Longo DL, Jameson JL, editors. Harrison’s principles of internal medicine. 16th ed. New York (NY): McGraw-Hill; 2005;1746-62.
2. Del Valle J, Scheiman JM. Zollinger-Ellison Syndrome. In: Yamada T, Alpers DE, Kaplowitz N, Laine L, Owyang C, Powell DW, eds. Textbook of Gastroenterology. Vol 1. 4th ed. Philadelphia, PA: Lippincott, Williams, and Wilkins; 2003;1321-77.
3. Kadakia SC, Wong RK, Maydonovitch CL, Nelson NR, Henkin RI. Serum and tissue zinc concentrations in patients with endoscopic esophagitis. Dig Dis Sci 1992;37(4):513-6.
4. Watanabe T, Arakawa T, Fukuda T, Higuchi K, Kobayashi K. Zinc deficiency delays gastric ulcer healing in rats. Dig Dis Sci 1995;40(6):1340-4.
5. Mann NS, Mann SK, Brawn PN, Weaver B. Effect of zinc sulfate & acetylcysteine on experimental gastric ulcer: in vitro study. Digestion 1992;53(1-2):108-13.
6. Santos LH, Feres CA, Melo FH, Coelho MM, Nothenberg MS, Oga S, et al. Anti-inflammatory, antinociceptive and ulcerogenic activity of a zinc-diclofenac complex in rats. Braz J Med Biol Res 2004;37:1205-13.
7. Cho CH, Ogle CW, Wong SH, Koo MW. Effect of zinc sulphate on ethanol- and indomethacin- induced ulceration and changes in prostaglandin E2 histaine levels in the rat gastric glandular mucosa. Digestion 1985;32(4):288-95.
8. Bandyopadhyay B, Bandyopadhyay SK. Protective effect of zinc gluconate on chemically induced gastric ulcer. Indian J Med Res 1997;106:27-32.
9. Bandyopadhyay B, Banerjee P, Bhattacharya B, Bandyopadhyay SK. Serum zinc level: a possible index in the pathogenesis of peptic ulcer syndrome. Biochem Mol Biol Int 1995;36(5):965-72.
10. Erzinkian KL, Lukasheva MV, Gobedzhavshili SD, Trapkov VA, Galaeva EV. Zinc metabolism in duodenal ulcer. Ross Med Zh 1992;(5-12):14-5.
11. Troskot B, Simicevic VN, Dodig M, Rotkvic I, Ivankovic D, Duvnjak M. The protective effect of zinc sulphate pretreatment against duodenal ulcers in the rat. BioMetals 1997;10(4):325-29.
12. Gu GG, Yong DG, Geng BQ. Anti-gastric ulcer activity of zinc sulfadiazine in rats. Zhongguo Yao Li Xue Bao 1990;11(5):460-2.
13. Trapkov VA, Gobedzhashvili OD, Erzinkian KL. The anti-ulcer action of zinc sulfate in models of acute and chronic ulcerogenesis in the rat duodenum. Eksp Klin Farmakol 1996;59(6):26-8.
14. Rodríguez de la Serna A, Díaz-Rubio M. Multicenter clinical trial of zinc acexamate in the prevention of nonsteroidal antiinflammatory drug induced gastroenteropathy. Spanish Study Group on NSAID Induced Gastroenteropathy Prevention. J Rheumatol 1994;21(5):927-33.
15. Tagliati CA, Kimura E, Nothenberg MS, Santos SR, Oga S. Pharmacokinetic profile and adverse gastric effect of zinc-piroxicam in rats. Gen Pharmacol 1999;33(1):67-71.
16. Frommer DJ. The healing of gastric ulcers by zinc sulphate. Med J Aust 1975;2(21):793-6.
17. Rodrigues LE, Galle P, Siry P, Vinhaes A. The protective effect of zinc on gastric ulceration caused by ethanol treatment. Braz J Med Biol Res 1989;22(1):41-50.
18. Puscas I, Sturzu L, Búzás G. Effect of ZnSO4 upon gastric acid secretion and carbonic anhydrase. Int J Clin Pharmacol Ther Toxicol 1985;23(11):609-12.
19. Maret W, Jacob C, Vallee BL, Fischer EH. Inhibitory sites in enzymes: zinc removal and reactivation by thionein. Proc Natl Acad Sci U S A 1999;96(5):1936-40.
20. D’Souza RS, Dhume VG. Gastric cytoprotection. Indian J Physiol Pharmacol 1991;35(2):88-98.
HOME | SEARCH | CURRENT ISSUE | PAST ISSUES
Professional
Medical Publications
Room No. 522, 5th Floor, Panorama Centre
Building No. 2, P.O. Box 8766, Saddar, Karachi - Pakistan.
Phones : 5688791, 5689285 Fax : 5689860
pjms@pjms.com.pk